Early access devices
Early Access Devices is an optional way to set up Axon Body cameras, Flex 2 cameras and controllers, and docks as test devices that receive firmware updates before the general release of the updates. This gives you the opportunity to review and test the changes in your own environment on a small scale before full deployment.
If you choose this option, the admin will receive an email informing them about the upcoming early access firmware update. When the update is deployed, only devices you specified (up to 15 cameras and 15 docks) will receive the early firmware update.
Test your early access devices regularly to ensure the updates do not affect your current processes. Control access and deployment of these devices, since their firmware version will differ from your other devices.
If you find a problem with a firmware update during testing, please contact Axon technical support so we can troubleshoot with you.
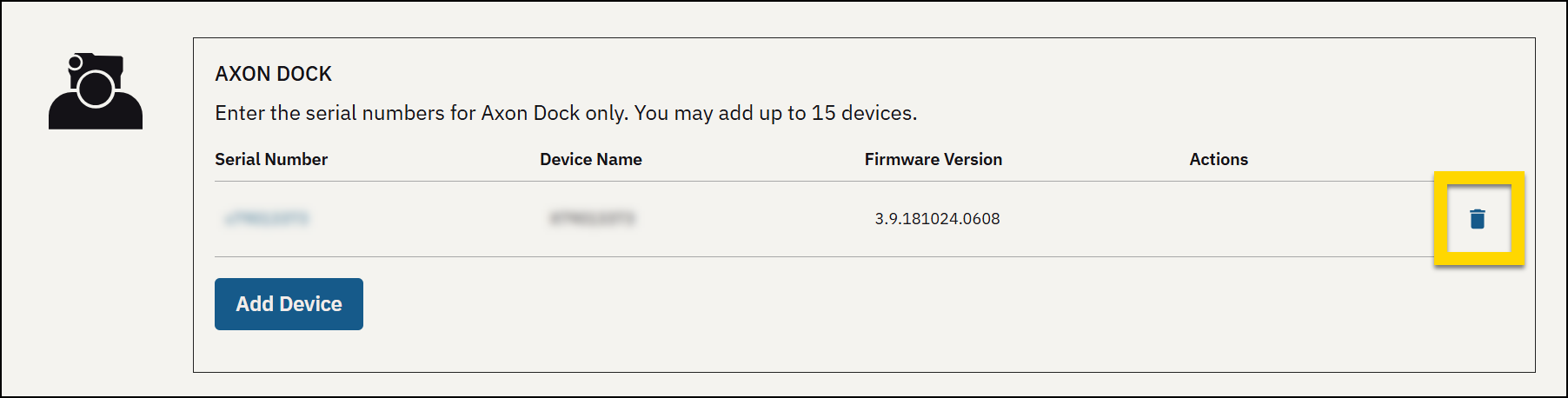
Add a device
- Select Admin.
- Under Devices and Applications, select Early Access Devices.
- Find the type of device you want to add, such as Axon Body 4 Camera, and select Add Device.
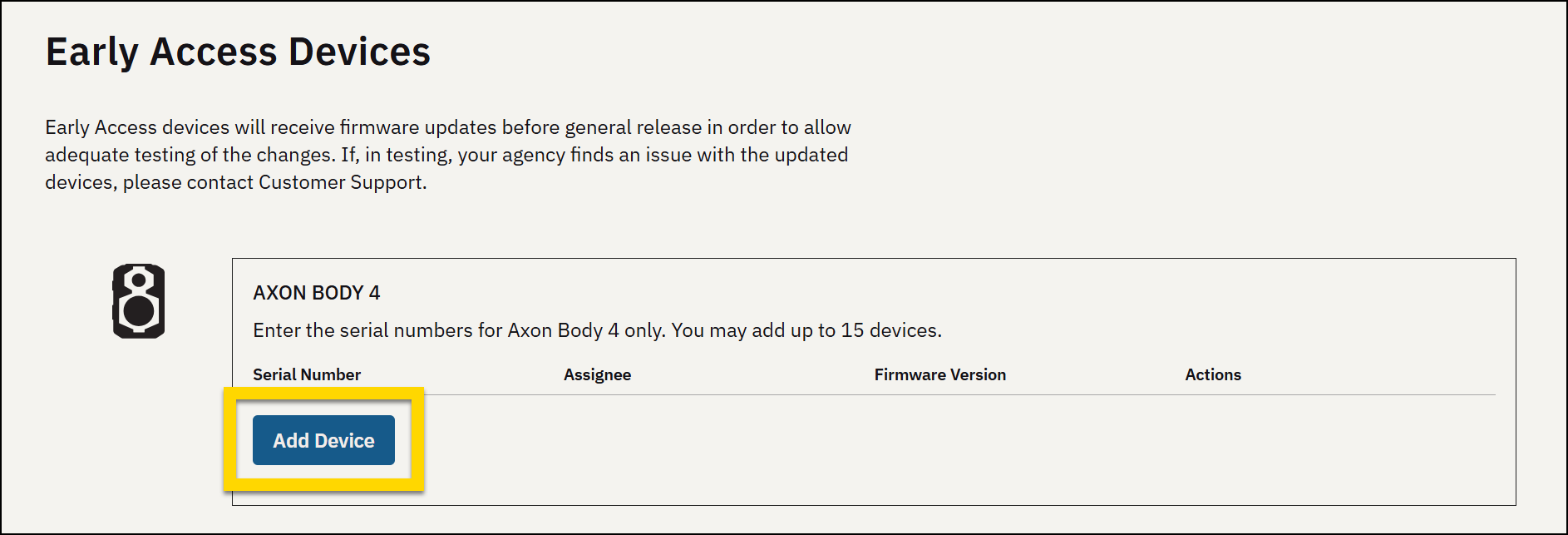
- Enter the device serial number.
- Select Save.